We use cookies to enhance your browsing experience and analyze site traffic. By continuing to use this site, you agree to our cookie policy. Privacy Policy
Epitope-Specific De Novo Antibody Design
Our Approach
No animal immunization needed. Directly generates novel antibodies targeting specified epitopes using three core AI models:AbGenerator generates diverse CDRH3 sequences,AffinityEvaluator predicts binding probability,AbEvolver simulates affinity maturation.
Validated Results
Why Is This Needed?
"Different binding epitopes lead to different efficacy and safety profiles. Current mainstream methods tend to target immunodominant regions, leaving cryptic or low-immunogenicity epitopes with scarce or zero candidates."
Key Advantages
Key Advantages
Difficult Epitopes
Traditional methods tend to target immunodominant regions. It is difficult to obtain effective antibodies for cryptic or conserved epitopes. Different epitopes can lead to different efficacy, safety, and mechanisms.
Lack of Precise Control
Current mainstream antibody discovery methods cannot guarantee obtaining target epitope antibodies. Candidate antibodies for low-immunogenicity, rare functional, or complex membrane protein targets are often scarce or absent.
Higher Design Difficulty Than General Proteins
CDRH3 conformations are highly flexible, antibodies lack rich co-evolutionary information, and antibody structure / complex data are significantly smaller, making design far more challenging than general protein binders.
Precise Epitope Targeting
The key advantage is precise control of antibody-epitope binding, avoiding the randomness of traditional immunization methods for rational "point-and-shoot" design.
Generative AI for CDRH3 Design
De novo CDRH3 design via generative models, breaking through limitations of template libraries and existing binder optimization paths, expanding the candidate sequence space.
Fully Human
Generated antibodies are fully human with reduced immunogenicity; all share the same light chain, avoiding heavy/light chain mispairing.
Multi-dimensional Scoring & Reports
Combines model predictions and structural features to score and filter candidates, outputting candidate sequences, rankings, and design reports for team review and experimental decisions.
Validation
Case Studies
Validated on multiple antigen targets. After designing 10,000+ candidate antibodies for specified epitopes, wet lab verification yielded multiple antibodies binding target epitopes with affinities ranging from micromolar to nanomolar.
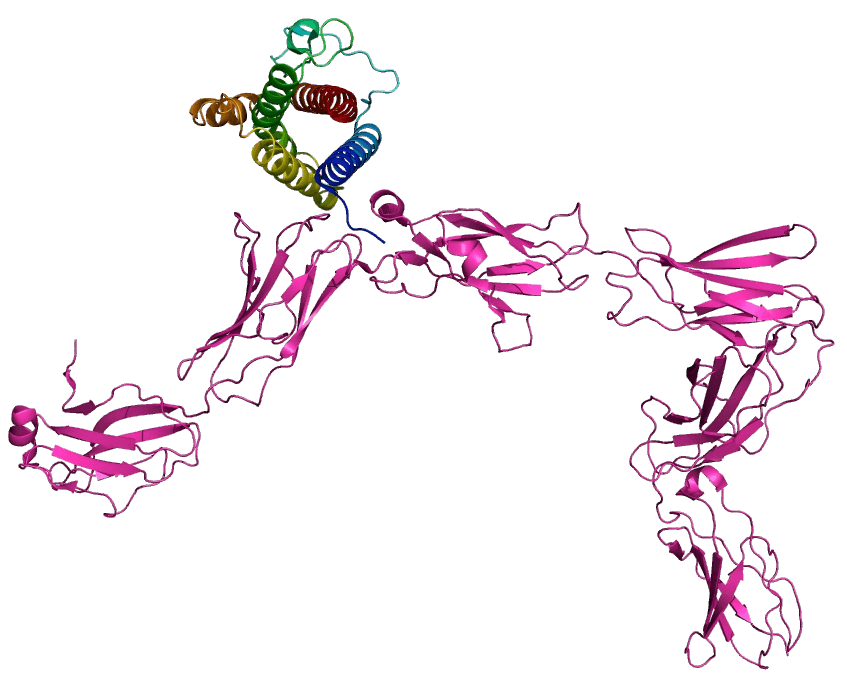
De novo design targeting a challenging functional epitope, yielding 7 BLI-validated positive antibodies
KD from 10-7 to 10-9 M; 2 antibodies blocked natural ligand binding with STAT-3 phosphorylation inhibition observed at the cellular level; cryo-EM further confirmed the precise epitope of P211858.

| Antibody | KD (M) | ka (1/Ms) | kd (1/s) |
|---|---|---|---|
| P95531 | 7.10E-07 | 2.01E+04 | 1.43E-02 |
| P211853 | 1.03E-07 | 1.39E+04 | 1.43E-03 |
| P211854 | 7.60E-09 | 8.50E+04 | 6.46E-04 |
| P211855 | 9.62E-08 | 9.14E+03 | 8.79E-04 |
| P211856 | 7.04E-08 | 9.20E+03 | 6.48E-04 |
| P211857 | 9.35E-08 | 9.46E+03 | 8.84E-04 |
| P211858 | 5.87E-09 | 9.68E+04 | 5.68E-04 |
| P211859 | 3.97E-09 | 1.70E+05 | 6.75E-04 |
| Sample | Value |
|---|---|
| P211858 | 159.5 nM |
| P211859 | 68.30 nM |
| Positive Control | 12.53 nM |
| Sample | Value |
|---|---|
| P95531 (PC) | 0.04748 |
| P211854 | 0.02238 |
| P211855 | 1.319 |
| P211856 | 0.6156 |
| P211857 | 2.490 |
| P211858 | 0.06618 |
| P211859 | 0.03155 |
| Sample | Value |
|---|---|
| P95531 (PC) | 9.840 |
| P211853 | ~ 0.2025 |
| P211854 | ~ 0.000 |
| P211855 | ~ 1.004e+029 |
| P211856 | ~ 0.000 |
| A083 (P211857) | ~ 0.1119 |
| P211858 | 0.2076 |
| P211859 | 0.1756 |
Pipeline
Pipeline
After the user specifies the epitope, antibody design, structure prediction, and screening are performed around it, ultimately delivering novel antibodies that meet expectations.
Experimental validation includes: ELISA / BLI / Epitope Competition / Cryo-EM / Cell-based Assays
Report
Report Example
Below are excerpts from a de novo antibody design service delivery report, showing the complete workflow from epitope analysis to candidate sequence evaluation.
Lab Guide
Wet Lab Validation Guide
Designed antibodies are trained on BioSiTech’s fully human shared light chain RenLite mice data. The report provides heavy chain HCDR3, heavy chain FR1-FR3, and shared light chain sequences. This guide helps you transition smoothly from computational results to experimental validation.
Gene Synthesis of Linearized scFv Fragments
Assemble the three-part sequences from the report into scFv: ① Heavy chain FR1-FR3 (10-20 matched frameworks recommended) + ② Heavy chain HCDR3 (choose from 10K/200K/1M tiers, oligopool synthesis) + ③ Heavy chain FR4-Linker-VK (shared light chain, fixed sequence).
Quality Control & Library Construction
Linearized scFv fragments require QC to ensure correct construction ratio ≥60%. Minor errors have limited impact on display screening.
Use phage display screening (yeast display also applicable). After 3+ rounds, assess enrichment via phage ELISA, then obtain positive binder sequences through monoclonal screening or NGS.
Recombinant Expression & Validation
Construct positive binder variable regions as full-length IgG (e.g. huIgG1/kappa), using CHO/HEK293 eukaryotic expression.
Binding Activity
Soluble antigens: ELISA / BLI / SPR; multi-pass membrane antigens: FACS with overexpressing cell lines or tumor cells.
Epitope Validation
Competition ELISA / Epitope Binning for rapid analysis; further confirmation via Cryo-EM / X-ray structural biology.
💡 Tip: During phage screening, set up a positive control library (mix known binders or Benchmark antibodies at 1:10000 ratio). If affinity improvement is needed, return directly to the platform for affinity maturation.
Use Cases
Use Cases and Deliverables
Best suited for projects with a defined functional epitope that need buildable candidates around that epitope.
Inputs to Prepare
What You Receive
What is the core advantage over traditional immunization screening?
Traditional methods cannot reliably hit a specified epitope, especially low-immunogenicity or rare functional epitopes. De novo design starts from the target epitope and uses generative AI to design CDRH3 and framework combinations, reducing uncertainty from random screening.
Why focus on CDRH3 design?
CDRH3 is critical for antigen recognition, but it is conformationally flexible and has limited structural data. Template-based or existing-binder optimization paths are constrained. The platform uses generative models to expand CDRH3 sequence space, giving epitope-specific projects more screenable candidates.
How do fully human and shared-light-chain designs help downstream development?
Fully human properties help reduce immunogenicity risk. Shared light chains reduce heavy/light-chain mismatch and downstream CMC complexity, making computational outputs easier to move into experimental validation.
How does multi-dimensional scoring help experimental decisions?
The report combines model predictions, structural features, and candidate ranking so teams can prioritize sequences more likely to hit the target epitope and fit library construction and screening, rather than receiving an unranked candidate pool.
Start AI-Driven De Novo Antibody Design
Starting from functional requirements, design novel antibodies around specified epitopes, breaking through the limitations of traditional methods.